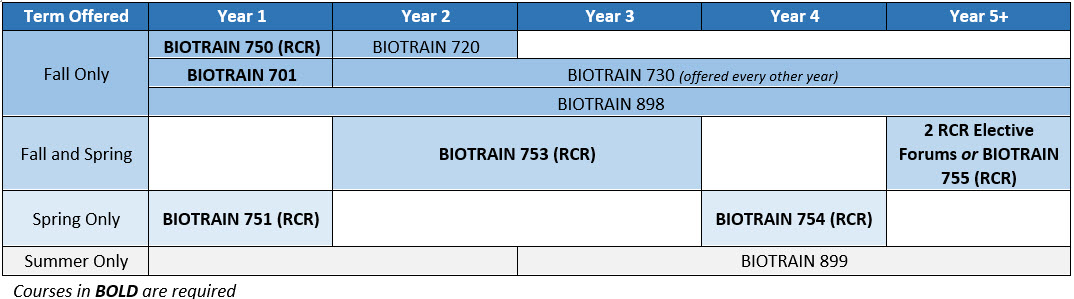
Responsible Conduct of Research (RCR) BIOTRAIN Courses
Academic integrity and research ethics are fundamental to the practice of science. We have created a rigorous program to train students in the highest standards for conducting research. All biomedical PhD students are required to participate in a total of 18 contact hours of in-person and online RCR courses. To receive credit for RCR training, students complete required assessments, and course credit is formally tracked by the university registrar to ensure that all RCR requirements are met prior to graduation. The following requirements apply to students that matriculated in Fall 2020 or later. If you matriculated prior to Fall 2020, transitional requirements are outlined here.
All first year biomedical PhD students will participate in BIOTRAIN 750, a one-day event prior to the start of classes in August. Topics covered will include conflict management, choosing a mentor, integrating well-being into graduate school, data management, identifying and reducing biases at Duke.
Course Availability: Prior to Fall semester (Orientation Week)
RCR Credit Hours: 4
Registration: Registration link sent via email to all incoming SOM students
Syllabus:
In the Spring, all first year students will participate in BIOTRAIN 751, a semester long course that utilizes online lectures/modules, in-person lectures and small group discussions, and focuses on Responsible Conduct of Research (RCR) and Rigor & Reproducibility (R&R) topics for early-stage graduate students. Each topic is accompanied by a short assessment and bi-monthly small group sessions centered on expanding the online/lecture material through discussion questions and case studies build community and encourage continual embedded dialogue about best practices in RCR and R&R. Small groups are led by faculty representing each of the PhD training programs and departments, with teaching assistance from senior graduate students and postdoctoral fellows.
Course Availability: Spring semester
RCR credit hours: 4
Registration: DukeHub
Syllabus:
At some point during the second or third year of the PhD program, every student must complete a self-paced, online BIOTRAIN 753. This course is offered via the Duke LMS (Learning Management System) in partnership with DOSI-ASIST and includes a set of 3 online interactive modules: Research Quality and Reproducibility, Data Management, and Data and Resource Sharing. The modules include interactive graphics, text-based activities, short videos, and discipline specific scenarios, and each module is accompanied by an assessment.
Course Availability: On demand
RCR credit hours: 2
Registration: Follow this link to the LMS system.
Note: These modules cannot be accessed using Internet Explorer.
Syllabus:
In the Spring of the 4th year, all students will participate in BIOTRAIN 754, a semester long course that utilizes online lectures/modules, in-person lectures and small group discussions, and focuses on Responsible Conduct of Research (RCR) and Rigor & Reproducibility (R&R) topics for advanced graduate students. Each topic is accompanied by faculty-led small group sessions centered on expanding the online/lecture material through discussion questions and case studies build community and encourage continual embedded dialogue about best practices in RCR and R&R, as well as a short assessment.
Course Availability: Spring semester
RCR credit hours: 4
Registration: DukeHub
Syllabus:
RCR Elective Forums: Students in years 5 and beyond will participate in two RCR elective forums. These are held throughout the year on a variety of topics to allow students to choose forums that relate to their personal interests and experience. These forums include PhD students from the humanities and social science and often provide a broader view of RCR than offered in previous training years. Each elective forum will provide 2 RCR credit hours.
Course Availability: Ongoing
RCR Credit Hours: 2 per forum
Registration: Through Duke Graduate School RCR Website
The Responsible Scientist Teaching Assistant/Peer Mentor: School of Medicine PhD students in years 4+ may earn BIOTRAIN 755 credit (replacing one of two required RCR Forums) by serving as a teaching assistant in BIOTRAIN 751: The Responsible Scientist I, and School of Medicine PhD students in years 3+ may earn up to two semesters of BIOTRAIN 755 credit (replacing up to two of two required RCR Forums) and a $1,000 stipend supplement by serving as an OBGE Graduate Student Peer Mentor over the course of one academic year, including participation in BIOTRAIN 701: Foundations in Professionalism and BIOTRAIN 751: The Responsible Scientist I.
RCR Credit Hours: 2
Registration: Apply through OBGE
Additional BIOTRAIN Courses
Besides RCR, BIOTRAIN offers a collection of preparatory courses specifically designed to build essential skills and knowledge for graduate students in the biomedical sciences throughout their PhD track.
This required six-week course equips first-year School of Medicine (SoM) biomedical PhD students with professionalism skills, including but not limited to stress management, effective communication, giving/receiving feedback, and mentor-mentee relationships. The course is team-taught by National Research Mentoring Curriculum-trained faculty members and Leadership and Management in Action Program-trained PhD students from diverse SoM PhD programs. Content includes interactive lectures that provide fundamental knowledge of key professional skills, and small group active learning sessions (Gateway Groups) during which students discuss and practice professionalism skills and receive feedback from faculty mentors, peer mentors, and peers. Course is available to SoM biomedical PhD students.
Course Availability: First six weeks of Fall semester
Course Credit: 1 Semester Hour
Registration: DukeHub
Syllabus:
Introduction to scientific grant writing for second- or third-year PhD students. This course contains lecture-based and active learning sessions. Content includes lectures combined with class discussions on grant agencies, format and structure of grant applications, concepts in peer review, best practices in articulating study design and data outcomes, rigor and reproducibility in a research plan, and crafting biological significance and training statements. Students write an NIH-style proposal and actively participate in topical study sections, typically comprising ~12 students and a faculty study section chair. Students receive oral and written critiques of their proposals from a Duke guest faculty reviewer and two peer study section members. This course introduces students to writing, critiquing, and submitting fellowships, while also preparing them for writing their preliminary exam document. Open only to second- or third-year students in School of Medicine biomedical PhD programs.
Course Availability: Fall semester
Course Credit: 3 Semester Hours
Registration: DukeHub
Syllabus:
Display of quantitative data is central to relaying results in scientific manuscripts, talks, and other visual communications. Learning to manage, display, communicate scientific results effectively and ethically is an important component of professional development for all scientists. This course will offer conceptual and hands-on training in data visualization for biomedical scientists, combining principles of graphic design with a deep dive into the Adobe suite of tools. In addition to using “out of the box” tools, the course will fully integrate coding in R and rigor and reproducibility best practices. Open only to students in School of Medicine PhD programs.
Course Availability: Every other year, typically Spring term (odd years)
Course Credit: 2 Semester Hours
Registration: DukeHub
This internship preparation course will serve as a companion course to BIOTRAIN 899 Internship and Experiential Learning to begin the process of allowing Biomedical PhD students to explore specializations and career paths outside academia and prepare them to make more informed career path decisions. The course will encourage students to plan a practical path for pursuing a career outside of academia, address benefits and challenges of participating in an internship during the PhD, and provide tools and skills to navigate the process of finding and successfully completing an internship as part of the PhD course of study.
Course Availability: Last six weeks of Fall and Spring semesters
Course Credit: 1 Semester Hour
Registration: DukeHub
Syllabus:
A majority of PhD-level biomedical trainees now choose non-academic-tenure-track careers after completion of the PhD (CBE Life Sci Educ. 2018 Spring; 17(1): ar16.). This internship course will allow Biomedical PhD students to explore specializations and career paths outside academia and prepare them to make more informed career path decisions. They will have an opportunity to experience non-academic work settings, build their career networks, gain additional mentorship, enhance leadership, communication, and teamwork skills, and explore interests and specializations not available to them in their on-campus training.
Prerequisite: BIOTRAIN 898 Gateway to Internship and Experiential Learning
Course Availability: Summer term only
Course Credit: 1 Semester Hour
Registration: T3 Professional Development Tab
BIOTRAIN 899 - STUDENT GUIDE - 2026
BIOTRAIN 899 - CHECKLIST - 2026
BIOTRAIN 899 - FACULTY GUIDE - 2026